Difference between revisions of "Neutron"
From Conservapedia
(See nucleon.) |
DavidB4-bot (Talk | contribs) (→External links: HTTP --> HTTPS, replaced: http://www.upi.com → https://www.upi.com) |
||
| (12 intermediate revisions by 10 users not shown) | |||
| Line 1: | Line 1: | ||
| − | [[Image:310px-Beta Negative Decay svg.png|right|thumb|300px|The | + | [[Image:310px-Beta Negative Decay svg.png|right|thumb|300px|The Feynmann diagram of a neutron decaying ([[beta decay|beta minus decay]])]] |
| − | A '''neutron''' is the neutral particle in an [[atom]]'s [[nucleus]]. It is | + | A '''neutron''' is the neutral particle in an [[atom]]'s [[nucleus]]. It is a [[baryon]], comprised of two down [[quark]]s and an up [[quark]]. It keeps the positive protons in the nucleus from being pushed apart from each other by their like charges. A lack or excess of neutrons causes an atom to become unstable; [[beta decay|beta radiation]] is the conversion of a neutron into a [[proton]] which emits an [[electron]] and an [[anti-electron neutrino]]. Although neutrons comprise a large part of all matter, they are inherently unstable if not bound to a nucleus, and undergoes beta minus decay in a brief period of time, with a half life of about 15 minutes.<ref>https://www.nist.gov/news-events/news/2014/02/toward-new-precision-measuring-neutron-lifetime</ref> |
| − | See | + | ==See also== |
| + | *[[Atom]] | ||
| + | *[[Proton]] | ||
| + | *[[Electron]] | ||
| + | *[[Periodic table of the elements]] | ||
| + | *[[Quark]] | ||
| + | *[[nucleon]] | ||
| + | |||
| + | ==References== | ||
| + | {{Reflist}} | ||
| + | |||
| + | ==External links== | ||
| + | |||
| + | *[https://www.upi.com/NewsTrack/Science/2007/09/18/study_contradicts_neutron_theory/8801/ Study contradicts neutron theory] | ||
| − | |||
| − | |||
[[Category:Physics]] | [[Category:Physics]] | ||
| + | [[Category:Subatomic Particles]] | ||
Latest revision as of 14:19, April 3, 2019
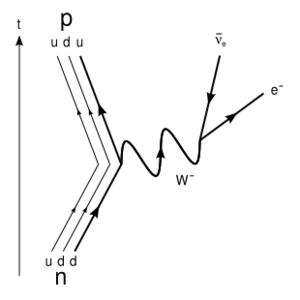
The Feynmann diagram of a neutron decaying (beta minus decay)
A neutron is the neutral particle in an atom's nucleus. It is a baryon, comprised of two down quarks and an up quark. It keeps the positive protons in the nucleus from being pushed apart from each other by their like charges. A lack or excess of neutrons causes an atom to become unstable; beta radiation is the conversion of a neutron into a proton which emits an electron and an anti-electron neutrino. Although neutrons comprise a large part of all matter, they are inherently unstable if not bound to a nucleus, and undergoes beta minus decay in a brief period of time, with a half life of about 15 minutes.[1]